
Meet the Authors
Key Takeaways
-
The shift towards AI-enabled delivery models in SAP implementations allows teams to accelerate timelines while maintaining essential regulatory compliance and documentation integrity, impacting life sciences organizations that require strict adherence to Good Practice (GxP) controls.
-
By optimizing the implementation lifecycle into distinct work categories—human-driven, human-led with AI support, and AI-led with human curation—companies can enhance efficiency and streamline processes without sacrificing the rigor needed for validation and control, benefitting SAP consultants and stakeholders.
-
Faster SAP S/4HANA delivery is achievable under disciplined management and the use of industry-specific accelerators that standardize repeatable processes, which ultimately helps optimize project timelines while ensuring compliance, impacting regulated industries where validation is critical.
Speed has always been the promise, and often the risk, in SAP programs. Compressing timelines can reduce costs and accelerate benefits realization, but it can also expose governance gaps, weaken documentation, and push critical decisions into late-stage testing. What is changing now is not the pressure to move faster, but the emergence of AI-enabled delivery models that claim to reduce manual workload without reducing rigor and to the need to prove those claims in inspection-ready terms.
UST argues that this shift allows teams to shorten SAP S/4HANA delivery cycles while maintaining Good Practice (GxP) fitness, validation readiness, and data integrity outcomes, particularly when automation is applied to repeatable implementation artifacts and human expertise is reserved for design and business-critical decisions. The core proposition is simple: accelerate what should be standard and protect what must remain specific. In regulated environments, “specific” includes control ownership, risk decisions, and evidence expectations, not just business process variation.
From ‘More People’ to ‘Smarter Work Allocation’
Traditional implementation models often scale by adding headcount, which can expand documentation and testing capacity but also increases coordination overhead and cost. AI-enabled delivery models attempt a different approach by treating the implementation lifecycle as a portfolio of work types, then optimizing each category differently. The goal is not fewer controls, but fewer avoidable handoffs and less rework.
Explore related questions
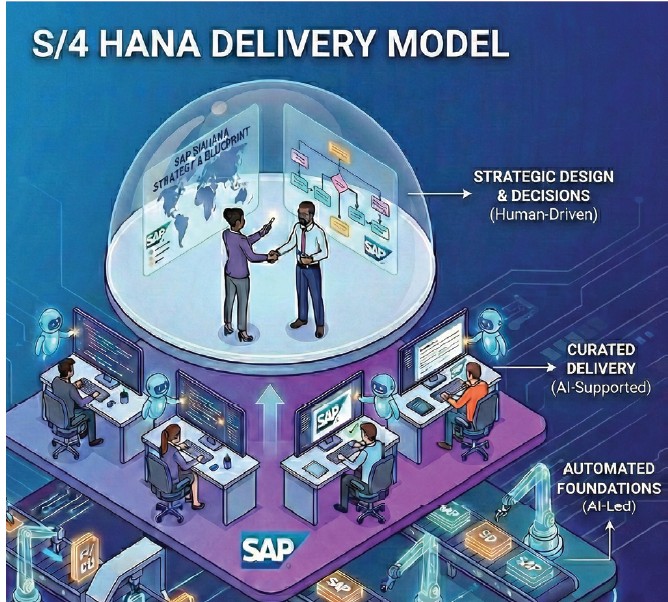
UST describes a delivery model that organizes SAP Activate work into three buckets:
- Human-driven work, where outcomes depend on business context and domain judgment. This includes design workshops, blueprint decisions, and the operating model choices that shape how the business will run in SAP S/4HANA. These activities are not candidates for full automation because they require stakeholder alignment, industry nuance, and trade-off decisions that determine value realization and control posture (e.g., where GMP decisions are made, who owns exceptions, and how evidence is generated).
- Human-led work with AI support, where consultants remain accountable but use automation to reduce manual effort and improve consistency. Examples include drafting and maintaining traceability matrices, updating process flows to reflect non-SAP integrations, and checking security configurations against requirements and controls. In this model, AI accelerates production and review, while consultants retain responsibility for accuracy and for documenting rationale when AI-generated outputs are accepted or overridden.
- AI-led work with human curation, where repeatable artifacts can be generated quickly but must be reviewed by experienced practitioners. In this model, activities such as requirements refinement, role-matrix generation, documentation production, and test content elements are automated first, then validated (reviewed, approved, versioned, and traceable) by subject matter experts to ensure accuracy and control.
The design intent is to reduce time spent producing artifacts and redeploy scarce expertise toward decisions that influence scope, adoption, controls, and measurable business outcomes. That intent is easiest to articulate and hardest to prove in regulated environments, where delivery models are tested not just on functional outcomes but on documentation, validation, and auditability.
UST frequently cites life sciences as proving ground for accelerated SAP delivery because of GxP controls, rigorous documentation standards, and computer systems validation (CSV) requirements that traditionally extend project timelines. UST’s position is that disciplined use of templates, prebuilt artifacts, and automation can shorten delivery cycles while preserving regulatory rigor.
Accelerating SAP S/4HANA Delivery
Faster SAP delivery claims are easiest to make in simple environments. They are hardest to substantiate in regulated industries, where validation, documentation, and auditability impose non-negotiable constraints. That is why life sciences frequently serves as a stress test for delivery models that promise speed without sacrificing control.
One example, according to UST, is a KalVista Pharmaceuticals SAP S/4HANA implementation delivered to support a fixed commercial launch deadline. The system was implemented as a validated, GxP-compliant environment within a 33-day delivery window. The reported scope included multiple plants and company codes, core finance integrated with Sage, procurement and materials management, planning and subcontracting, batch management and traceability, quality status management, inventory, and sales.
Timelines of this nature are achievable only under specific conditions: disciplined scope control, extensive reuse of prebuilt assets, tightly governed validation processes, and sustained business availability throughout the delivery cycle. The relevance of the example lies not in its duration, but in what it illustrates about where acceleration is possible when regulatory rigor is treated as a design constraint rather than an afterthought. One mechanism that enables this kind of discipline is the use of industry accelerators.
Industry accelerators are often misunderstood as one-size-fits-all solutions. In practice, their value lies in standardizing what should be repeatable, while preserving flexibility where regulatory or business differentiation demands it.
In life sciences, this typically includes preconfigured process flows, predefined user requirements, and traceability structures. These are aligned to regulatory expectations such as the FDA’s 21 CFR Part 11, EU Annex 11 (where applicable) risk-based initial security role designs that account for segregation-of-duties controls, and validation documentation frameworks that support computer systems validation. Training and change assets are also increasingly delivered in a partially complete state, allowing programs to start from a governed baseline rather than a blank page.
Used correctly, these accelerators do not replace design work. They reduce rework. The cycle-time benefit comes from eliminating the need to recreate artifacts that are already well understood, while ensuring changes are traceable from requirements through configuration and test evidence and that deviations are documented with rationale and approvals.
Redefining What ‘Faster’ Actually Means
Accelerated delivery is rarely driven by a single tool. It emerges when tools are orchestrated within a coherent delivery operating model. Components in modern SAP programs include process analysis platforms such as SAP Signavio, enterprise architecture tools like LeanIX, test automation solutions including Tricentis, data migration platforms such as Syniti, digital adoption tooling like WalkMe, and SAP Business Technology Platform (BTP) for integration and extension.
Individually, these tools are not differentiators. Many programs deploy similar stacks and still struggle. The differentiator is whether the toolchain is embedded into governance: whether artifacts flow consistently across tools, whether automation outputs are reviewed and validated, and whether changes are controlled as scope evolves. Without that discipline, tools add complexity rather than reducing risk.
Speed in SAP programs is often framed as a headline metric. A more helpful definition focuses on outcomes. Acceleration is meaningful only if it reduces cycle time while preserving or improving quality, compliance, and adoption readiness.
In practice, this means being explicit about what changes. Which activities move from manual to automated? Where does defect density decrease, and where does it not? What controls ensure that automation does not propagate errors at scale? How is change managed so that faster builds do not result in delayed adoption or increased post-go-live remediation?
Acceleration that compresses timelines without addressing these questions shifts risk rather than removing it.
Discovery Before Acceleration
Effective acceleration does not begin with execution. It begins with understanding reality. Process maturity, customization levels, data readiness, regulatory exposure, and organizational capacity all shape what can be accelerated safely.
Structured discovery—combining process and system analysis, architectural assessment, and roadmap definition—establishes the constraints within which acceleration is viable. Without that baseline, compressed timelines tend to expose issues late, when remediation is most expensive.
Faster technical delivery raises, rather than lowers, the importance of change discipline. When build and test cycles compress, adoption debt accumulates quickly if learning, communication, and readiness do not keep pace.
Role-based training, adoption telemetry, and readiness checkpoints must move in lockstep with configuration and validation. Otherwise, accelerated programs risk surfacing problems after go live, including workarounds, control failures, and unrealized benefits.
AI-enabled delivery can materially change how SAP programs are executed, but only when applied with clear boundaries. Repeatable artifacts can be automated. Design accountability, validation decisions, and control ownership cannot. AI can accelerate evidence production; it cannot own evidence.
For SAP transformation leaders, the takeaway is not to pursue headline timelines. It is to demand delivery models that show where automation is applied, how compliance and validation are protected, and how adoption is managed alongside speed. Acceleration succeeds not when programs move faster, but when they arrive with fewer unresolved risks and an inspection-ready evidence package.




